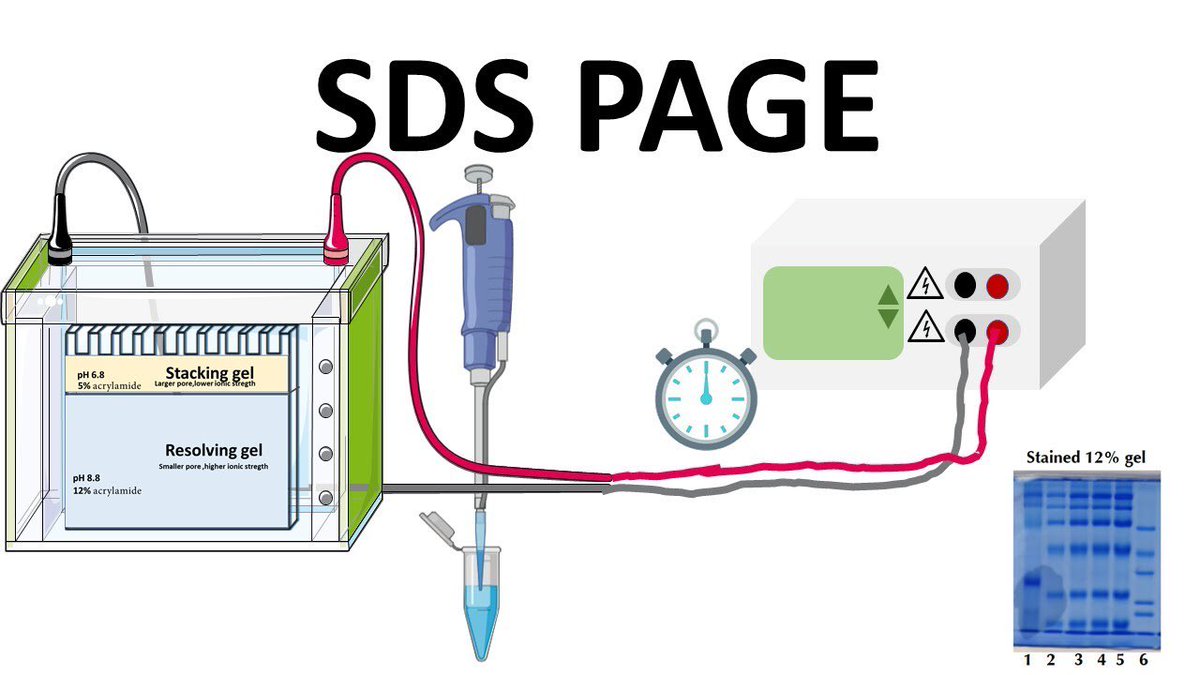
Check that bubbles are emerging from the electrode at the bottom of the apparatus. Attach the lid and plug the leads into the power source.Be sure to record how your gel was loaded. Below is a sample of how you and your neighboring group might load your gel. Add the volume slowly and watch to ensure that it sinks to the bottom of the well. Carefully load 10µL of ladder and 20µL of each prepared sample into the wells of the gel using gel-loading tips.Pour the Running buffer into the outer chamber until it is half-full. Pour buffer into the inner chamber of the gel box between the gels so that the wells are completely submerged. Remove the sealing tape from the bottom of the gel before loading. Two gels can be run simultaneously in each apparatus. 
Prepare samples for the gel by mixing 10μL of the CL, FT, W, and E fractions with 30 μL of Wash buffer and 10μL of 5x SDS loading buffer.1 Mini-PROTEAN 1D gel electrophoresis system/power source.Fractions from purification (lysate, protein elution).All the samples will also be run in duplicate on another gel – one gel will be stained for TOTAL protein with Coomassie and the other gel will be used for a Western blot to identify a SPECIFIC protein with an antibody. We will run a gel of our fractions from the nickel column purification to identify the GFP-containing fraction and assess its purity.įour groups will run their samples from the His 6 -GFP purification on the same gel. Because SDS is negatively charged, each protein will be separated by size, similar to the separation of different DNA fragments in an agarose gel. Dodecylsulfate (SDS) is a detergent that binds to and denatures (unfolds) proteins. Sodium dodecylsulfate polyacrylamide gel electrophoresis (SDS-PAGE) is used to separate and visualize individual proteins from a complex mixture.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
